Calprotectin is a complex of the mammalian proteins S100A8 and S100A9.[1][2] Other names for calprotectin include MRP8-MRP14, calgranulin A and B, cystic fibrosis antigen, L1, 60BB antigen, and 27E10 antigen.[2][3] The proteins exist as homodimers but preferentially exist as S100A8/A9 heterodimers or heterotetramers (calprotectin) with antimicrobial, proinflammatory and prothrombotic properties.[4][5] In the presence of calcium, calprotectin is capable of sequestering the transition metals iron,[6] manganese and zinc[2][7] via chelation.[8] This metal sequestration affords the complex antimicrobial properties.[2][7] Calprotectin is the only known antimicrobial manganese sequestration protein complex.[9] Calprotectin comprises as much as 60% of the soluble protein content of the cytosol of a neutrophil,[2][10][11] and it is secreted by an unknown mechanism during inflammation.[3] Faecal calprotectin has been used to detect intestinal inflammation (colitis or enteritis) and can serve as a biomarker for inflammatory bowel diseases.[10][12] Blood-based calprotectin (in serum and plasma) is used in diagnostics of multiple inflammatory diseases, including autoimmune diseases, like arthritis, and severe infections including sepsis.[13][14]
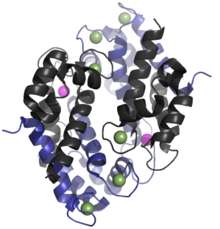
The human homologue of calprotectin is a 24 kDa dimer,[9] and is formed by the protein monomers S100A8 (10,835 Da) and S100A9 (13,242 Da).[4][5] The primary structure of calprotectin can vary between species. For instance, the mouse homologue of S100A8 is 10,295 Da,[15] while the S100A9 homologue is 13,049 Da.[16] Early size exclusion chromatography experiments incorrectly indicated that calprotectin had a molecular mass of 36.5 kDa;[2][11] occasionally this value is used in contemporary literature. Calprotectin S100A8-S100A9 dimers can non-covalently pair with one another to form 48 kDa tetramers.
Calprotectin has a high affinity for calcium, zinc, iron, and manganese.[10][11][17][6] Each of S100A8 and S100A9 contain two EF-hand type Ca2+ binding sites,[9][3] and calprotectin is able to bind a total of four calcium ions per dimer or eight calcium ions per tetramer.[18] Calcium binding induces a conformational change in the complex that improves its affinity for transition metals, and promotes tetramer formation.[2][9] A maximum of two transition metal ions may bind to each calprotectin S100A8-S100A9 dimer.[9]
A calprotectin dimer can bind only one manganese or iron ion with high affinity, and it can do this only in the presence of calcium.[9][19][6] Zinc can bind at two sites within the calprotectin dimer, and this can occur in the absence of calcium.[2] Calcium, however, improves calprotectin's affinity for zinc.[9] While calprotectin metal binding occurs at the interface of S100A9 and S100A8 monomers, the independent monomers have some capacity for zinc binding, and may contribute to zinc homeostasis within mammals.[2][4][5]

The first of the two calprotectin metal binding sites consists of a His3Asp motif, with S100A8 contributing two histidine ligands (His83 and His87), and S100A9 contributing a histidine and an aspartic acid ligand (His20 and Asp30).[9] The second site can coordinate metals through a tetra-histidine (His4) or a hexa-histidine (His6) binding motif. In the case of His4 binding, S100A8 coordinates through both His17 and His27 while S100A9 coordinates through His91 and His95.[9] In hexa-histidine binding two further histidine residues, His103 and His105, are recruited from the C-terminal end of S100A9 to enable octahedral coordination of the transition metal.[9] Manganese or iron are bound by the calprotectin dimer at this His6 site.[9][6] Zinc can be bound to either of the sites that form at the interface between S100A8 and S100A9 monomers.[9][19]
Calprotectin constitutes up to 60% of soluble protein content in the cytosol of neutrophil granulocytes,[2][10][11] and it can be found at a lower concentration in monocytes, macrophages, and squamous epithelial cells.[2][10][11] Calprotectin enters into pus and abscess fluid during neutrophil cell death, along with other antimicrobial proteins.[2]
Mammalian cells secrete calprotectin during the inflammatory response. Circulating activated platelets and platelet-leukocyte aggregates are increased in acute and chronic sterile thrombo-inflammatory diseases. Plasma calprotectin is elevated in persons with metabolic syndrome, a disease characterized by chronic inflammation.[20] Calprotectin is secreted in the mouth during inflammation of the gingiva and during oral candidiasis infection.[21][22] People who have mutations in the calprotectin gene appear susceptible to serious gum infections.[21] Manganese sequestration by calprotectin is likely important during lung inflammation.[7] The exact mechanism by which S100A8 and S100A9 is secreted by mammalian cells during inflammation remains unknown.[3] In lung autopsies from patients with inflammation caused by COVID-19, heterodimeric S100A8/A9 is mainly detected in neutrophils and deposited on vessel walls.[23] Platelet glycoprotein Ib alpha (GP1BA;GPIbα) is the receptor for S100A8/A9 on platelets.[23] In vitro, platelets adhere to and partially spread on S100A8/A9, leading to the formation of distinct populations of P-selectin+ and phosphatidylserine+ platelets. The prothrombotic pathway initiated by interaction of S100A8/A9 with GPIbα induces the formation of procoagulant platelets and fibrin (CD36 has a supporting role).[23]
Transition metals are essential to the survival of all organisms.[24] Mammals strictly limit metal availability as a part of the innate immune system, and this helps prevent infection by microbes and fungi.[24] Calprotectin was first described in the 1980s as a mammalian antimicrobial protein that acts through the sequestration of zinc.[1][2][9] It is now known that calprotectin also has antibacterial and antifungal properties that arise from its ability to sequester manganese and iron.[7][9][6] Calprotectin is the only known antimicrobial agent that acts through manganese sequestration.[9]
Calprotectin becomes available in the intestinal lumen via leukocyte shedding,[1] active secretion,[2][11] cell disturbance, and cell death.[1][11] This results in elevated faecal calprotectin levels, which can be detected in the stool.[1][11] Elevated faecal calprotectin levels therefore indicate migration of neutrophils into the intestinal mucosa, which occurs during intestinal inflammation.[1][11][17] As people with active inflammatory bowel diseases (IBD) such as ulcerative colitis or Crohn disease have as much as a 10-fold increase in faecal calprotectin levels,[10] the measurement of faecal calprotectin can serve as a biochemical test for these diseases.
Although a relatively new test, faecal calprotectin is regularly used as an indicator for IBD during treatment and as a diagnostic marker.[12] Faecal calprotectin tests can also function in distinguishing patients with irritable bowel syndrome from those with IBD.[1][11] Calprotectin is useful as a marker, as it is resistant to enzymatic degradation, and can be easily measured in faeces.[25] Although faecal calprotectin correlates significantly with disease activity in people with confirmed IBD,[26] elevated faecal calprotectin can be a false-positive indicator of IBD under some conditions. Importantly, intake of proton pump inhibitor is associated with significantly elevated calprotectin values.[27] Furthermore, positive faecal calprotectin does not help in localizing IBD, or in distinguishing ulcerative colitis from Crohn's disease.[1] Faecal calprotectin can also indicate other gastrointestinal conditions such as colorectal cancer, gastroenteritis, and food intolerance.[1] Calprotectin levels vary depending on age, comorbidity, and may vary day-to-day within individuals.[1] Faecal calprotectin could be used as a preliminary screen in otherwise functional patients suspected of having IBD, or as a means of following mucosal healing.[1] In patients with SARS-CoV-2 infection, elevated faecal calprotectin has been demonstrated to correlated with COVID-19 induced thrombosis even in patients without gastrointestinal symptoms.[28] The potential for using faecal calprotectin in this way is debated, however, and cut-off levels have not been agreed upon.[1]